PPT Unit 3 PowerPoint Presentation, free download ID5685070
And you have one more electron to worry about. And so that electron would go into a 3S orbital. So the full electron configuration is 1S2, 2S2, 2P6, and 3S1. When I want to figure out how many valence electrons sodium has, the number of valence electrons would be equal to the number of electrons in the outermost shell, the outermost energy level.

How to find Valency? What are valence electrons? Teachoo (2023)
Created by video journalist Brady Haran working with chemists at The University of Nottingham. Element Neon (Ne), Group 18, Atomic Number 10, p-block, Mass 20.180. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images.
Electron Dot Diagram Periodic Table
Get ready to find out how many valence electrons neon has! In this video, we'll explore the fascinating world of chemistry and discover why valence electrons.
How Many Valence Electrons Are in the Neon Family
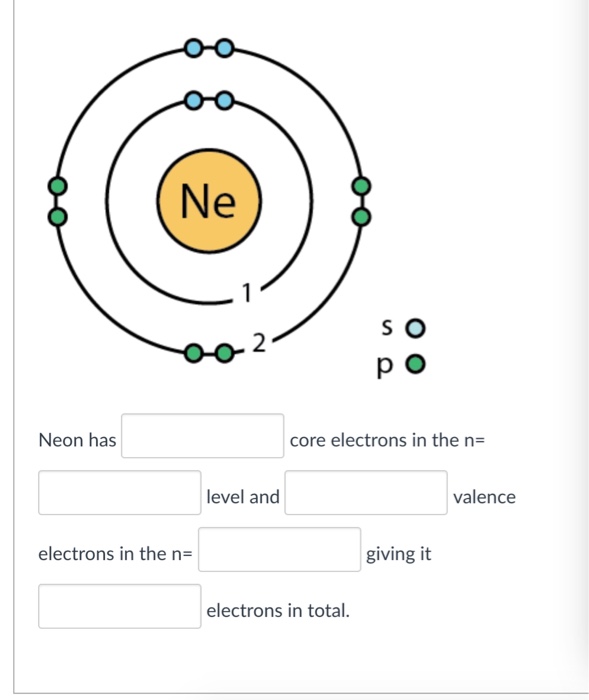
Step-3: Determine the valence shell and calculate the total electrons. The third step is to diagnose the valence shell (orbit). The last shell after the electron configuration is called the valence shell. The total number of electrons in a valence shell is called a valence electron. The electron configuration of neon shows that the last shell.
Periodic table of elements with valence electrons kcJuli
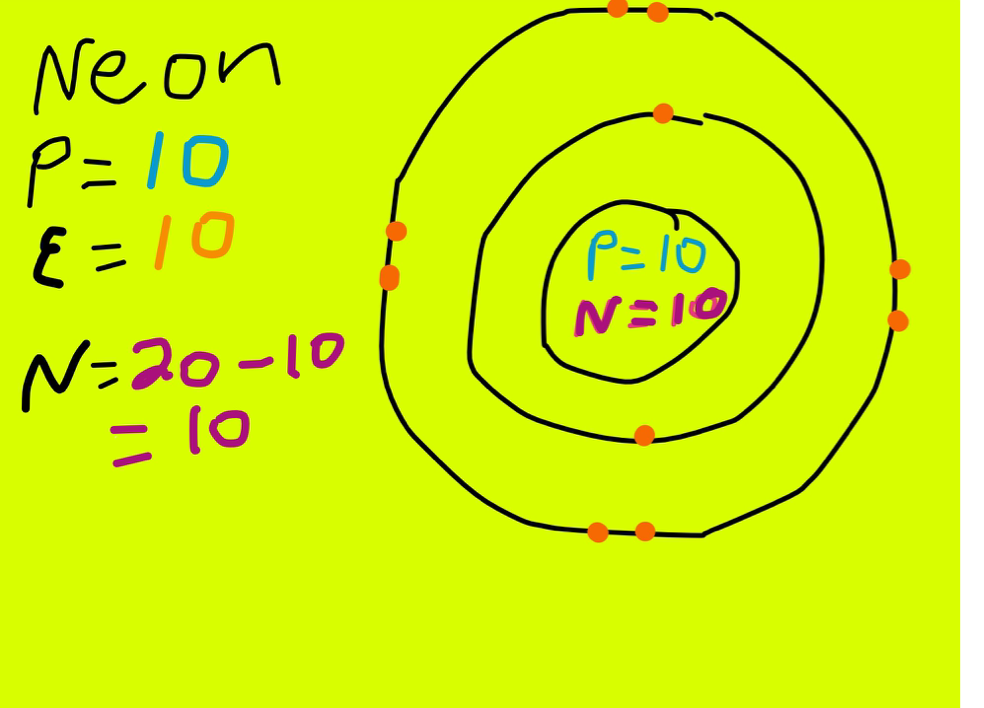
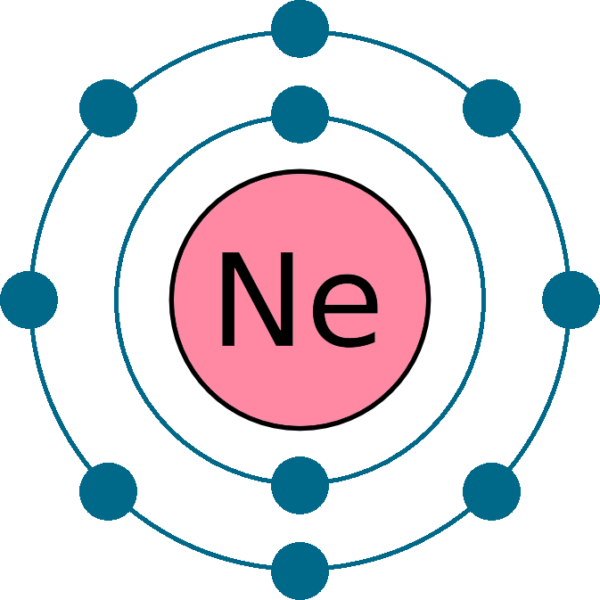
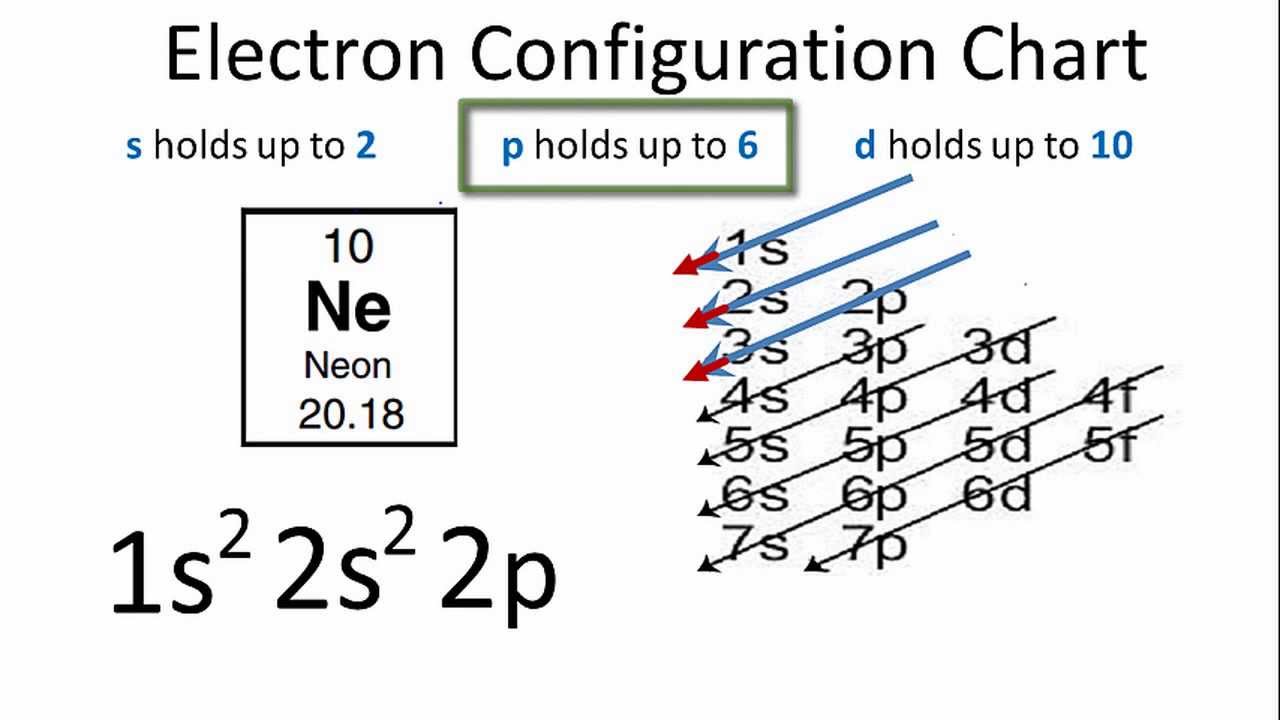
The electronic structure of neon (Ne) is 1s 2 2s 2 2p 6. This means that neon has two electrons in the 1s orbital, two electrons in the 2s orbital, and six electrons in the 2p orbitals. Neon has a completely filled valence shell, making it an inert or noble gas.

How to find Valency? What are valence electrons? Teachoo
The neon atom has a total of 10 electrons so, we have to put 10 electrons in orbitals. the first two electrons will go in the 1s orbital as the S orbital can hold a maximum of 2 electrons only. Again another two electrons will go in 2s orbital and the remaining 6 will go to the 2p orbital.
Electron Configuration Of Neon My XXX Hot Girl
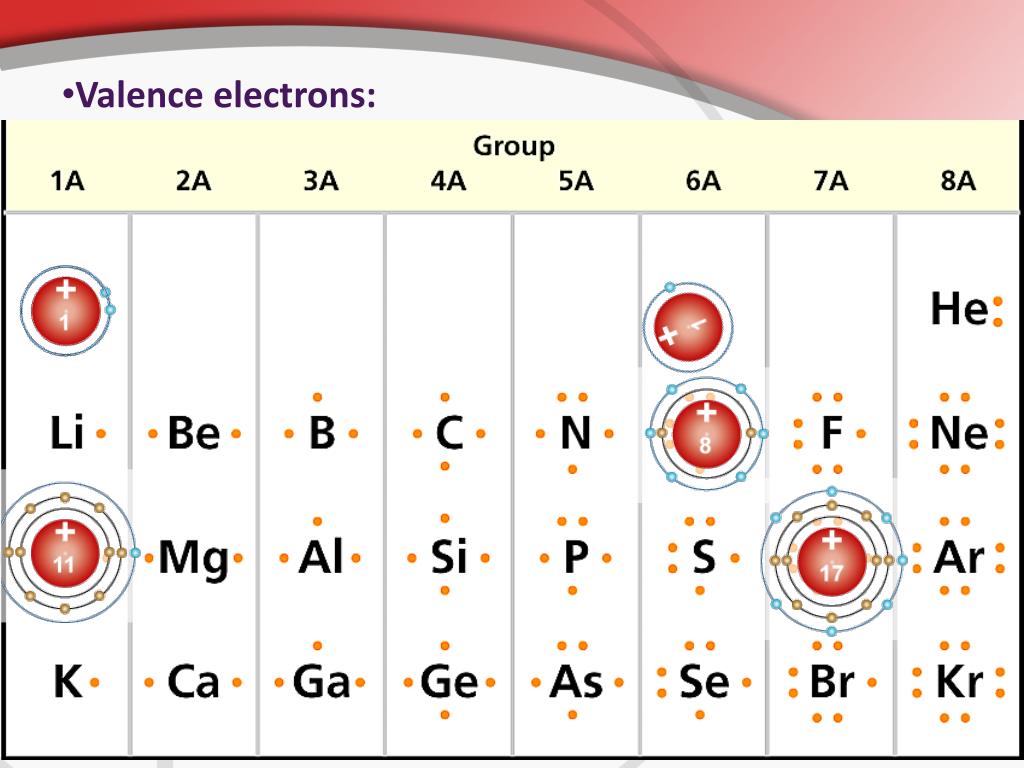
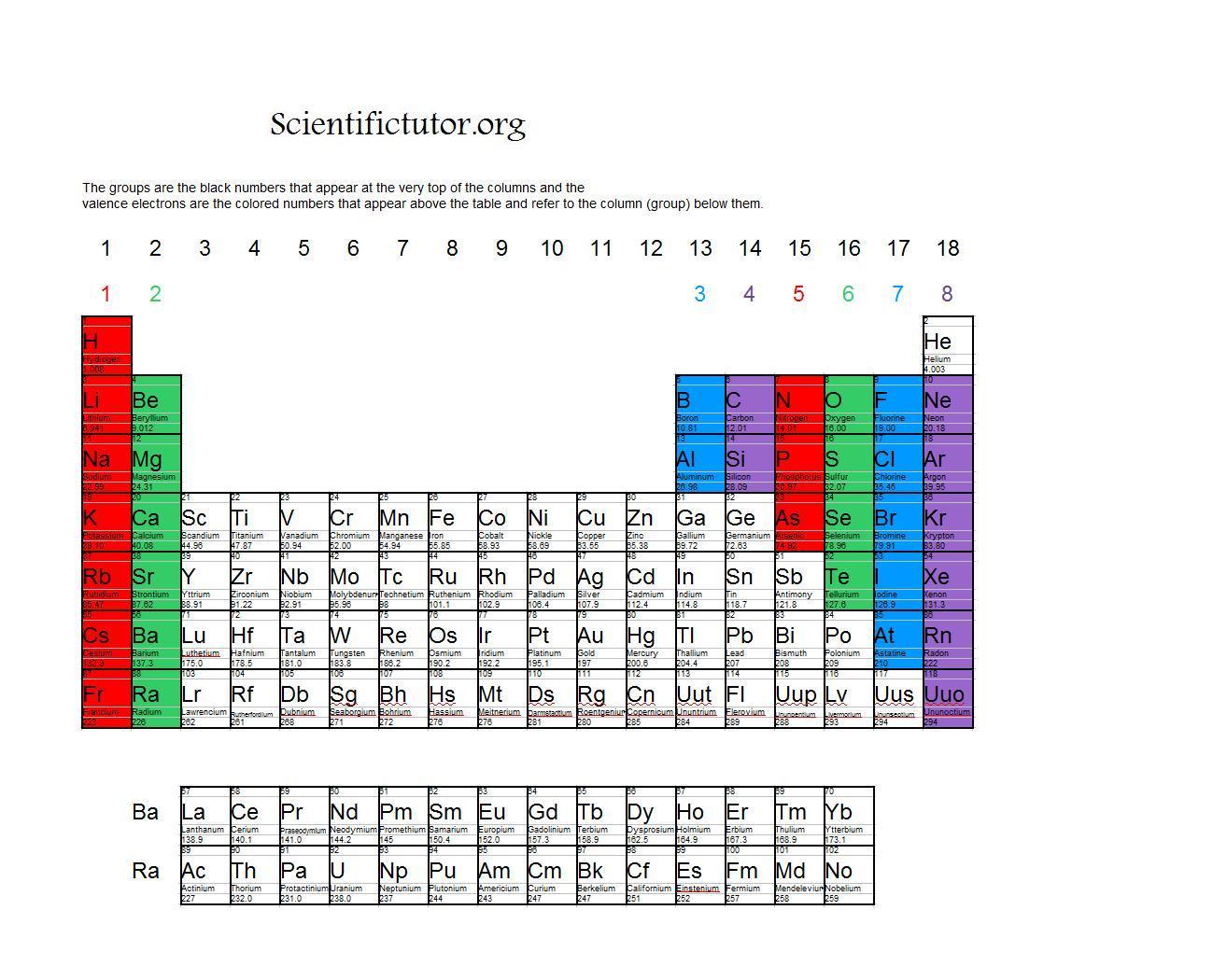
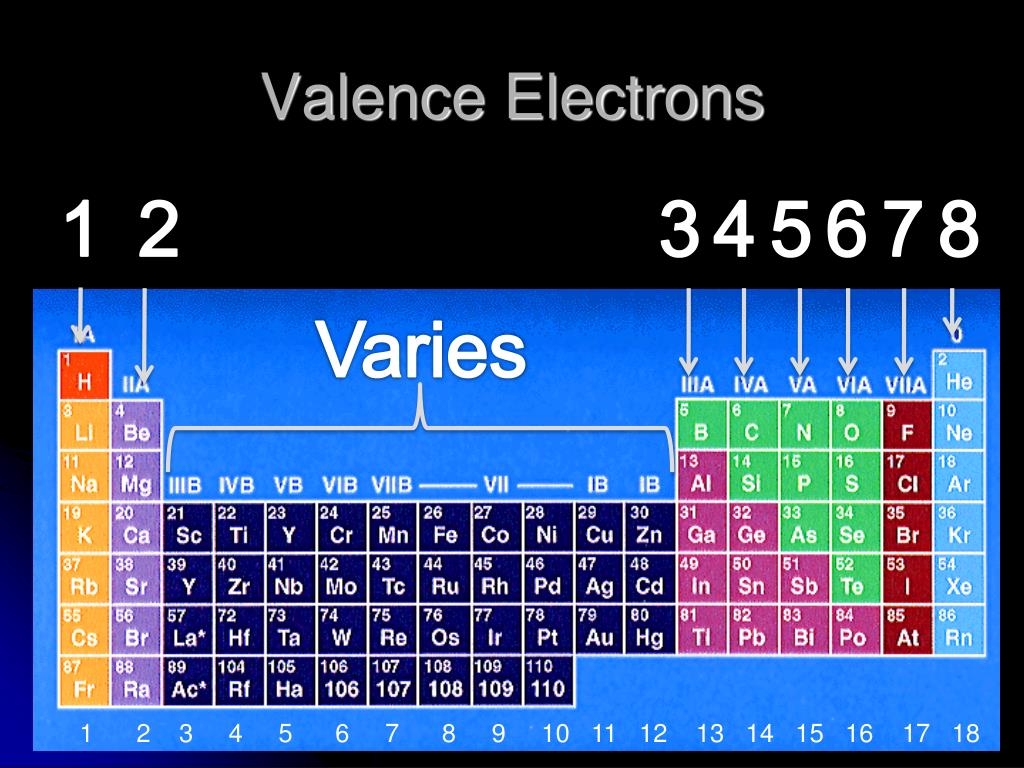
Each element has a number of valence electrons equal to its group number on the Periodic Table. Figure %: The periodicity of valence electrons This table. Helium (He) and Neon (Ne) have outer valence shells that are completely filled, so neither has a tendency to gain or lose electrons. Therefore, Helium and Neon, two of the so-called Noble.

How many valence electrons does neon(Ne) have?
For example, silicon is in Group IVA (Group 14), so each atom would have four valence electrons. Chlorine is in Group VIIA (Group 17), so it would have seven valence electrons. Calcium would have two valence electrons, since it is in Group IIA (Group 2). Helium is the only exception for the main group elements. The first energy level holds a.

Free Download Periodic Table Help Names Valence Electrons For Free Uploadto
Four covalent bonds.Carbon has four valence electrons and here a valence of four. Each hydrogen atom has one valence electron and is univalent. In chemistry and physics, valence electrons are electrons in the outermost shell of an atom, and that can participate in the formation of a chemical bond if the outermost shell is not closed. In a single covalent bond, a shared pair forms with both.

Neon Protons, Neutrons, Electrons Complete Guide
Valence electrons are the electrons present in the outermost shell of an atom. You can easily determine the number of valence electrons an atom can have by looking at its Group in the periodic table. For example, atoms in Groups 1 and 2 have 1 and 2 valence electrons, respectively. Atoms in Groups 13 and 18 have 3 and 8 valence electrons.
Electron Configuration Of Neon In Excited State worksheet today
Valence electrons: For main group elements (i.e s-block and p-block elements), the valence electrons are the electrons present in the outermost orbit. But for most of the transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core.

How many valence electrons does neon(Ne) have?
How many valence electrons does boron have? Recognize that the second principal energy level consists of both the \(2s\) and the \(2p\) sublevels, and so the answer is three.. Neon, with its configuration ending in \(2s^2 2p^6\), has eight valence electrons. Valence electrons for transition elements. Transition elements are a bit trickier.
Solved Ne 2 p o Neon has core electrons in the n level and
Any element in group 18 has eight valence electrons (except for helium, which has a total of just two electrons). Examples include neon (Ne), argon (Ar), and krypton (Kr). Oxygen, like all the other elements in group 16, has six valence electrons. Discuss further with Flexi.

What Are Valence Electrons And How To Find Them Where Are They Located
Explanation: Neon, Z = 10, has eight valence electrons. This closed shell configuration makes neon supremely difficult to oxidize, and difficult to reduce. The inertness, the lack of reactivity, of this Noble Gas, is a function of its electronic configuration. To which group of the Periodic Table does neon belong? Neon, Z=10, has eight valence.

Electron Configuration Chart With Orbitals
There are two ways to find the number of valence electrons in Neon (Ne). The first is to use the Periodic Table to figure out how many electrons Neon has in.
Elements Their Atomic, Mass Number,Valency And Electronic Configuratio
Every subshell has a # of orbits s/p/d/f that can each hold 2 electrons each (one has the opposite spin of the other). The first shell (of all atoms) has 1 subshell of s-orbitals containing 1 s orbital. This means that the first shell can hold 2 electrons. The second shell has 2 subshells: 1 s-orbital and 3 p-orbitals.